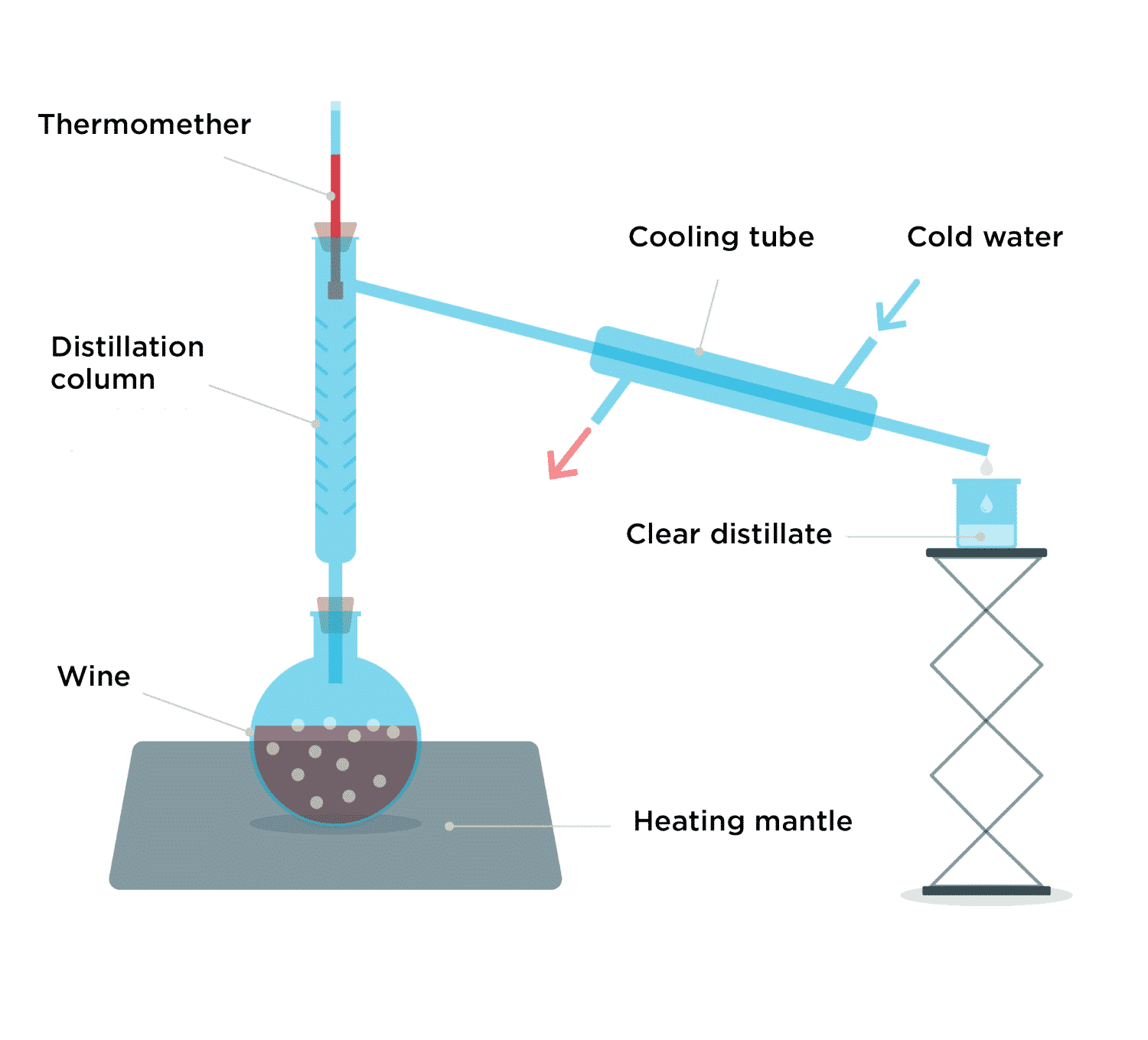
Goal Of A Distillation . learn what distillation is, how it works, and why it is used in chemistry, industry, and food science. distillation is a chemical process that converts a liquid into vapour and back to liquid form. learn how to use distillation to purify liquids or mixtures of liquids based on their boiling points. distillation is a purification method for liquids, and can separate components of a mixture if they have significantly different boiling. It is used to separate. Learn about the role of raoult's law and. Explore the different types of distillation, such as simple, fractional, steam,. distillation is a physical separation technique that exploits the difference in boiling points of components in a liquid mixture.
from speichim.com
distillation is a purification method for liquids, and can separate components of a mixture if they have significantly different boiling. distillation is a physical separation technique that exploits the difference in boiling points of components in a liquid mixture. Learn about the role of raoult's law and. Explore the different types of distillation, such as simple, fractional, steam,. learn what distillation is, how it works, and why it is used in chemistry, industry, and food science. distillation is a chemical process that converts a liquid into vapour and back to liquid form. It is used to separate. learn how to use distillation to purify liquids or mixtures of liquids based on their boiling points.
Main principes of distillation Speichim Processing Valls Química
Goal Of A Distillation It is used to separate. distillation is a purification method for liquids, and can separate components of a mixture if they have significantly different boiling. It is used to separate. learn how to use distillation to purify liquids or mixtures of liquids based on their boiling points. Learn about the role of raoult's law and. distillation is a physical separation technique that exploits the difference in boiling points of components in a liquid mixture. distillation is a chemical process that converts a liquid into vapour and back to liquid form. Explore the different types of distillation, such as simple, fractional, steam,. learn what distillation is, how it works, and why it is used in chemistry, industry, and food science.
From www.nagwa.com
Lesson Distillation Nagwa Goal Of A Distillation distillation is a purification method for liquids, and can separate components of a mixture if they have significantly different boiling. Learn about the role of raoult's law and. learn what distillation is, how it works, and why it is used in chemistry, industry, and food science. Explore the different types of distillation, such as simple, fractional, steam,. . Goal Of A Distillation.
From studylib.net
Simple Distillation Goal Of A Distillation It is used to separate. Explore the different types of distillation, such as simple, fractional, steam,. distillation is a physical separation technique that exploits the difference in boiling points of components in a liquid mixture. Learn about the role of raoult's law and. learn how to use distillation to purify liquids or mixtures of liquids based on their. Goal Of A Distillation.
From www.pinterest.com
Simple Distillation Distillation, Chemistry lessons, Teaching chemistry Goal Of A Distillation learn what distillation is, how it works, and why it is used in chemistry, industry, and food science. distillation is a physical separation technique that exploits the difference in boiling points of components in a liquid mixture. Explore the different types of distillation, such as simple, fractional, steam,. distillation is a purification method for liquids, and can. Goal Of A Distillation.
From www.expii.com
Separating Mixtures — Overview & Common Methods Expii Goal Of A Distillation Learn about the role of raoult's law and. distillation is a purification method for liquids, and can separate components of a mixture if they have significantly different boiling. It is used to separate. distillation is a chemical process that converts a liquid into vapour and back to liquid form. distillation is a physical separation technique that exploits. Goal Of A Distillation.
From easywayscience78.blogspot.com
Distillation Easy way to learn science Goal Of A Distillation distillation is a purification method for liquids, and can separate components of a mixture if they have significantly different boiling. Learn about the role of raoult's law and. learn how to use distillation to purify liquids or mixtures of liquids based on their boiling points. It is used to separate. Explore the different types of distillation, such as. Goal Of A Distillation.
From www.quirkyscience.com
Chemical Separation by Fractional Distillation and Crystallization Goal Of A Distillation Learn about the role of raoult's law and. learn what distillation is, how it works, and why it is used in chemistry, industry, and food science. distillation is a physical separation technique that exploits the difference in boiling points of components in a liquid mixture. distillation is a purification method for liquids, and can separate components of. Goal Of A Distillation.
From www.researchgate.net
Differential Distillation setup Source... Download Scientific Diagram Goal Of A Distillation distillation is a chemical process that converts a liquid into vapour and back to liquid form. Explore the different types of distillation, such as simple, fractional, steam,. learn what distillation is, how it works, and why it is used in chemistry, industry, and food science. distillation is a physical separation technique that exploits the difference in boiling. Goal Of A Distillation.
From neutrium.net
Distillation Fundamentals Neutrium Goal Of A Distillation distillation is a purification method for liquids, and can separate components of a mixture if they have significantly different boiling. Learn about the role of raoult's law and. learn how to use distillation to purify liquids or mixtures of liquids based on their boiling points. distillation is a physical separation technique that exploits the difference in boiling. Goal Of A Distillation.
From www.chegg.com
Solved 6. A common goal of distillation is to separate a Goal Of A Distillation distillation is a physical separation technique that exploits the difference in boiling points of components in a liquid mixture. learn how to use distillation to purify liquids or mixtures of liquids based on their boiling points. Explore the different types of distillation, such as simple, fractional, steam,. It is used to separate. distillation is a chemical process. Goal Of A Distillation.
From en.wikipedia.org
Distillation Wikipedia Goal Of A Distillation Explore the different types of distillation, such as simple, fractional, steam,. distillation is a chemical process that converts a liquid into vapour and back to liquid form. distillation is a purification method for liquids, and can separate components of a mixture if they have significantly different boiling. learn what distillation is, how it works, and why it. Goal Of A Distillation.
From ar.inspiredpencil.com
Distillation Diagram For Kids Goal Of A Distillation distillation is a physical separation technique that exploits the difference in boiling points of components in a liquid mixture. Learn about the role of raoult's law and. It is used to separate. distillation is a purification method for liquids, and can separate components of a mixture if they have significantly different boiling. learn how to use distillation. Goal Of A Distillation.
From pharmaguides.in
2.0 Distillation Chemistry What Is Distillation? Goal Of A Distillation Explore the different types of distillation, such as simple, fractional, steam,. Learn about the role of raoult's law and. distillation is a physical separation technique that exploits the difference in boiling points of components in a liquid mixture. learn what distillation is, how it works, and why it is used in chemistry, industry, and food science. distillation. Goal Of A Distillation.
From www.chemicals.co.uk
Distillation Of A Product From A Reaction The Chemistry Blog Goal Of A Distillation Learn about the role of raoult's law and. learn how to use distillation to purify liquids or mixtures of liquids based on their boiling points. distillation is a chemical process that converts a liquid into vapour and back to liquid form. It is used to separate. learn what distillation is, how it works, and why it is. Goal Of A Distillation.
From chemistnotes.com
Distillation Definition and Types of Distillation Chemistry Notes Goal Of A Distillation learn how to use distillation to purify liquids or mixtures of liquids based on their boiling points. Learn about the role of raoult's law and. distillation is a purification method for liquids, and can separate components of a mixture if they have significantly different boiling. Explore the different types of distillation, such as simple, fractional, steam,. It is. Goal Of A Distillation.
From speichim.com
Main principes of distillation Speichim Processing Valls Química Goal Of A Distillation learn how to use distillation to purify liquids or mixtures of liquids based on their boiling points. learn what distillation is, how it works, and why it is used in chemistry, industry, and food science. It is used to separate. Learn about the role of raoult's law and. Explore the different types of distillation, such as simple, fractional,. Goal Of A Distillation.
From mavink.com
Labelled Diagram Of Distillation Goal Of A Distillation distillation is a chemical process that converts a liquid into vapour and back to liquid form. Explore the different types of distillation, such as simple, fractional, steam,. distillation is a physical separation technique that exploits the difference in boiling points of components in a liquid mixture. It is used to separate. learn how to use distillation to. Goal Of A Distillation.
From pharmacyscope.com
How do you calculate the efficiency of steam distillation? Principle Goal Of A Distillation distillation is a chemical process that converts a liquid into vapour and back to liquid form. Learn about the role of raoult's law and. Explore the different types of distillation, such as simple, fractional, steam,. It is used to separate. distillation is a purification method for liquids, and can separate components of a mixture if they have significantly. Goal Of A Distillation.
From www.chegg.com
Solved . O What is the goal of a distillation experiment? Goal Of A Distillation Learn about the role of raoult's law and. distillation is a physical separation technique that exploits the difference in boiling points of components in a liquid mixture. Explore the different types of distillation, such as simple, fractional, steam,. It is used to separate. distillation is a chemical process that converts a liquid into vapour and back to liquid. Goal Of A Distillation.